
Welcome to Amanda's NICU Education




Hi! My name is Amanda. I'm a NICU nurse, Clinical Nurse Specialist, NICU Educator... basically your NICU BFF. If you want to talk NICU, I'm here for you! I love everything about NICU nursing and I'm eager to learn and share my knowledge with all my NICU friends.
I have been a NICU nurse since 2009 I am currently a Clinical Nurse Specialist in a Level IV NICU in Los Angeles.
I am passionate about educating the next generation of NICU nurses. I share my knowledge through platforms such as Instagram and Facebook and am excited to have you here on my website!
Click on the button below to sign up for my newsletter filled with NICU education and tips for all experience levels.

Not very many people love taking tests but as a self-acclaimed "forever student" who has taken (and passed) five different certification exams I am no longer afraid of tests! "Way to brag", you might be thinking but I want to help YOU pass your certification exam too!
Introducing Amanda's RNC-NIC Success digital course - your ultimate study companion!
Gain unlimited, on-demand access for life, ensuring you're primed to ace your certification exam.
I'm here to help you succeed and I can't wait for you to share with me that you PASSED the RNC-NIC EXAM!!!










NEC Awareness Month
NEC Awareness Month:
What NICU Nurses Need to Know About the Latest Research in Necrotizing Enterocolitis
Necrotizing enterocolitis (NEC) is one of the diagnoses that every NICU nurse remembers. It can begin subtly… some emesis, increased abdominal girth, a baby who "just doesn't look right" and progress rapidly into bowel necrosis, sepsis, surgery, and lifelong complications.
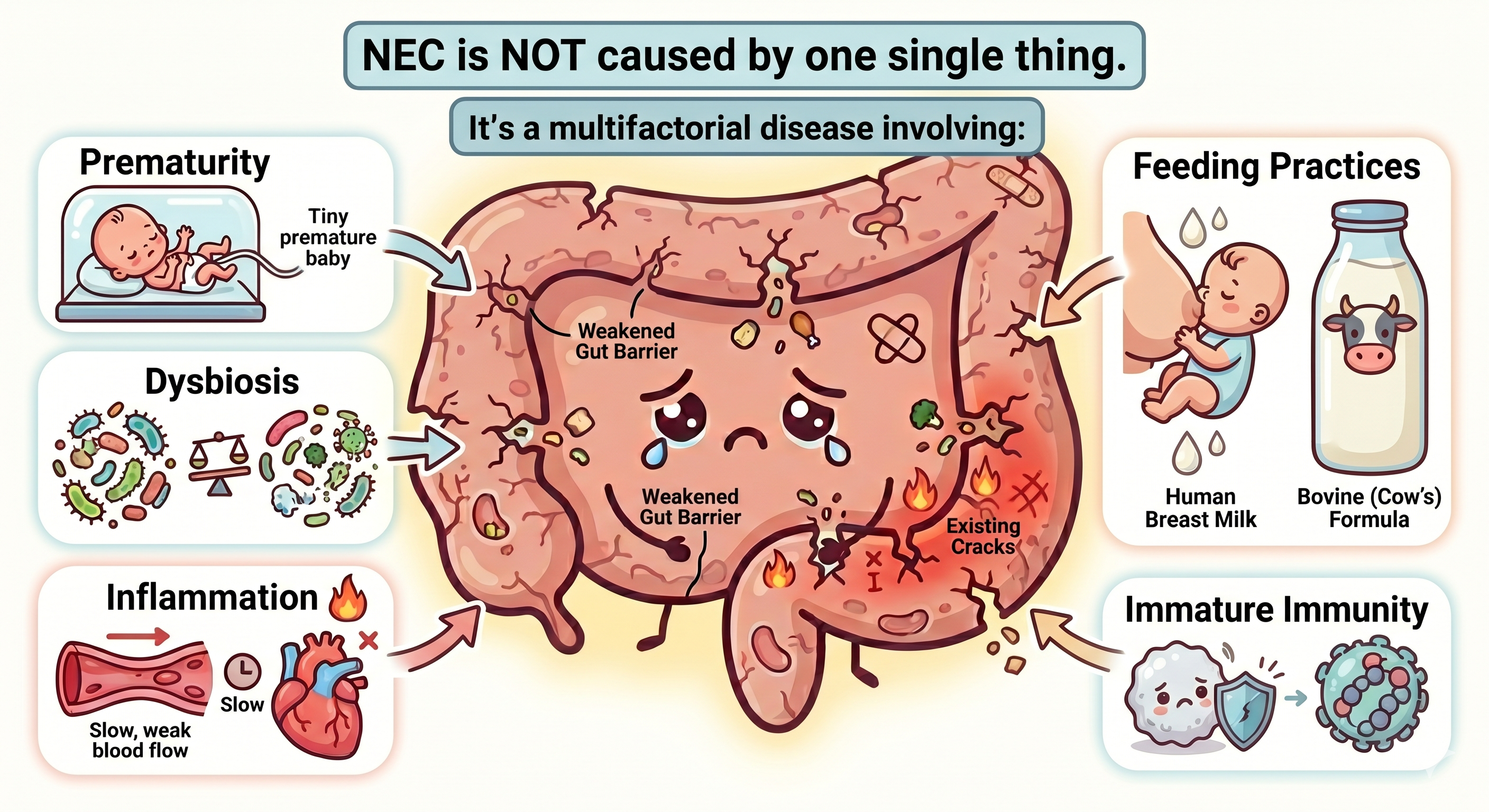
Even with advances in neonatal care, NEC remains one of the leading causes of morbidity and mortality in premature infants. Recent research continues to reinforce something many NICU nurses already know at the bedside: NEC is not caused by one single thing. It is a multifactorial disease involving intestinal immaturity, inflammation, dysbiosis, altered perfusion, feeding practices, and an exaggerated immune response in the vulnerable preterm gut.
May is NEC Awareness Month: I just returned from the ONE conference, where I had the opportunity to speak with nurses about protecting the newborn microbiome. We talked about which microbes matter in early neonatal life, what happens when microbial balance is disrupted, and why the metabolites are emerging as a significant piece of the NEC puzzle. It’s an important conversation that I want to share with you, so I have woven some of those concepts throughout this blog.
Let's break down some of the most important and evolving concepts NICU nurses should know.
NEC Is More Than "A Gut Disease"
It's important that we recognize that NEC is a systemic inflammatory disease. The intestinal injury is devastating, and the downstream effects extend far beyond the bowel.
Survivors may experience:
Neurodevelopmental impairment
White matter injury
Growth failure
Intestinal failure and short bowel syndrome
Feeding difficulties
Long-term respiratory and developmental complications
Recent reviews emphasize that systemic inflammation and sepsis likely contribute to many of these long-term outcomes. These babies often require long-term multidisciplinary support and neurodevelopmental follow-up. The disease you help prevent or detect early at the bedside has implications that extend years into a child's life.
The Premature Intestine Is Uniquely Vulnerable
Research continues to support prematurity as the single greatest risk factor for NEC.
The premature intestine has:
Increased intestinal permeability ("leaky gut")
Immature motility
Reduced digestive enzymes
Altered blood flow regulation
Reduced mucus protection
Immature immune defenses
Altered microbiome development
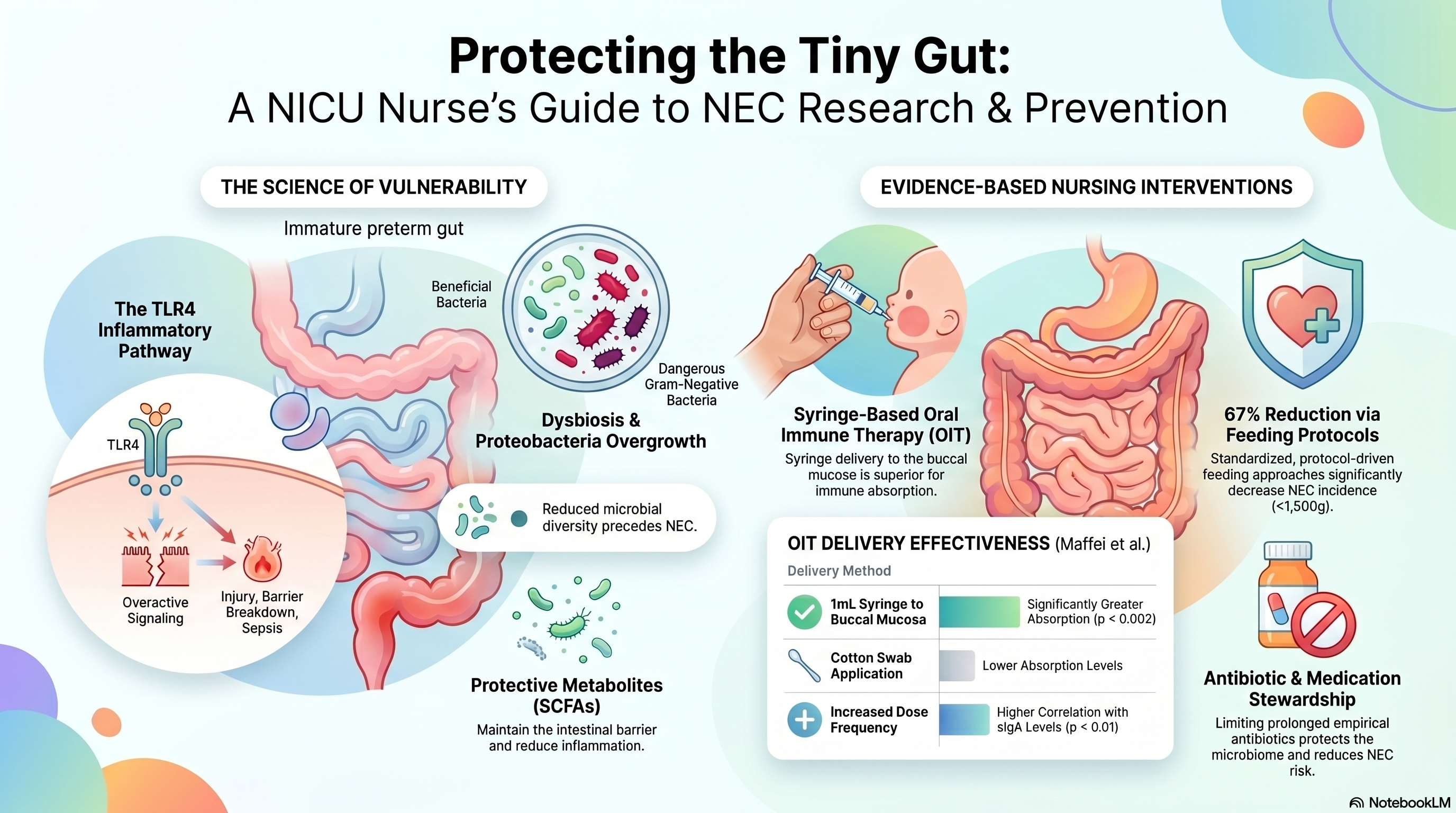
The result is an intestine that is highly vulnerable to injury and exaggerated inflammation. One of the major pathways being studied is Toll-Like Receptor 4 (TLR4) signaling. TLR4 is part of the innate immune system. In premature infants, TLR4 activity appears exaggerated. When activated by certain bacterial products, it can trigger:
Enterocyte injury
Barrier breakdown
Vasoconstriction
Ischemia
Cytokine release
Bacterial translocation
Sepsis
Researchers now consider TLR4-driven inflammation one of the central mechanisms in NEC pathogenesis and it is one of the active targets for future therapies. This includes TLR4 inhibitors and anti-inflammatory biologics currently being studied in preclinical settings.
The Microbiome: What We Now Know (And Why It Matters at the Bedside)
At the ONE conference, one of the themes was how much the field of neonatal microbiome science has shifted our understanding of disease. NEC is at the center of that conversation.
Over the last few years, NEC research has increasingly focused on the gut microbiome, and the findings are reshaping how we think about prevention.
What the Research Shows
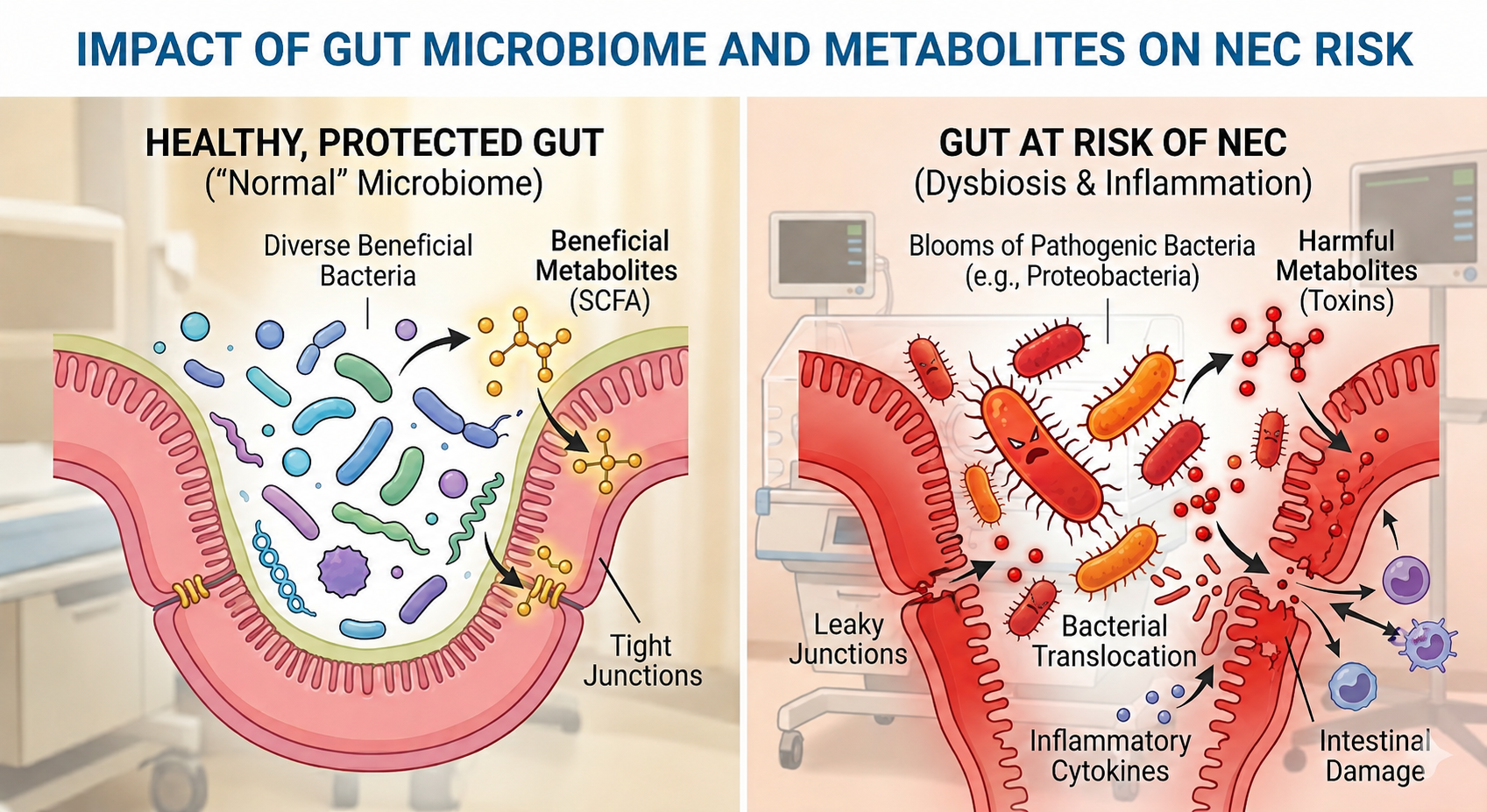
Many studies show that infants who develop NEC often demonstrate:
Reduced microbial diversity in the days and weeks before clinical disease
Overgrowth of Proteobacteria: gram-negative bacteria like Klebsiella, Enterobacter, and E. coli. These are common in the NICU environment and particularly dangerous in the preterm gut
Dysbiosis prior to clinical disease, meaning microbial imbalance may be detectable before NEC fully declares itself
Altered bile acid metabolism, which worsens mucosal damage and fuels further inflammation through specific signaling pathways
Altered short-chain fatty acid (SCFA) production
Let’s talk a little more about SCFA….
Short-Chain Fatty Acids: Why the Metabolites Matter
Short-chain fatty acids are produced when beneficial gut bacteria ferment dietary fiber and other substrates. They include compounds like butyrate, propionate, and acetate, and they play critical roles in:
Maintaining intestinal barrier integrity
Regulating immune responses
Reducing inflammation
Supporting enterocyte health
In the preterm infant, dysbiosis means less SCFA production. And less SCFA production means a more vulnerable gut lining, more inflammation, and greater NEC risk. This is one reason researchers are actively studying SCFA supplementation as a potential preventive strategy.
Similarly, bile acid dysregulation (another metabolite disruption seen in pre-NEC microbiome profiles) can worsen mucosal damage through inflammatory signaling cascades. Research on Bacteroides fragilis (a beneficial commensal) has shown it may help restore bile acid balance and reduce NEC-like injury in animal models, though this is still preclinical.
What This Means at the Bedside
Understanding the microbiome helps explain why our evidence-based prevention practices work.
Human milk directly shapes the preterm microbiome! It supports beneficial bacteria, limits pathogen overgrowth, and provides human milk oligosaccharides (HMOs) that fuel protective microbes
Antibiotic stewardship is microbiome protection! Every course of antibiotics reshapes which organisms survive in the preterm gut
Minimizing unnecessary antacid/acid-suppressing medications also matters, as these alter the gut environment in ways that promote dysbiosis
Skin-to-skin care supports microbial colonization from the parent's skin, contributing to healthier microbiome development
The bedside practices you already know are, at a molecular level, microbiome-protection strategies.
Human Milk Remains One of the Most Protective Interventions We Have
Despite all the cutting-edge biomarker and molecular research happening in NEC, one of the strongest protective interventions remains beautifully simple: human milk. Exclusive human milk diets continue to show significant protection against NEC.
Human milk provides:
Immunologic protection
Human milk oligosaccharides (HMOs) that selectively feed beneficial bacteria
Anti-inflammatory bioactive compounds
Microbiome-shaping effects
Improved intestinal barrier function
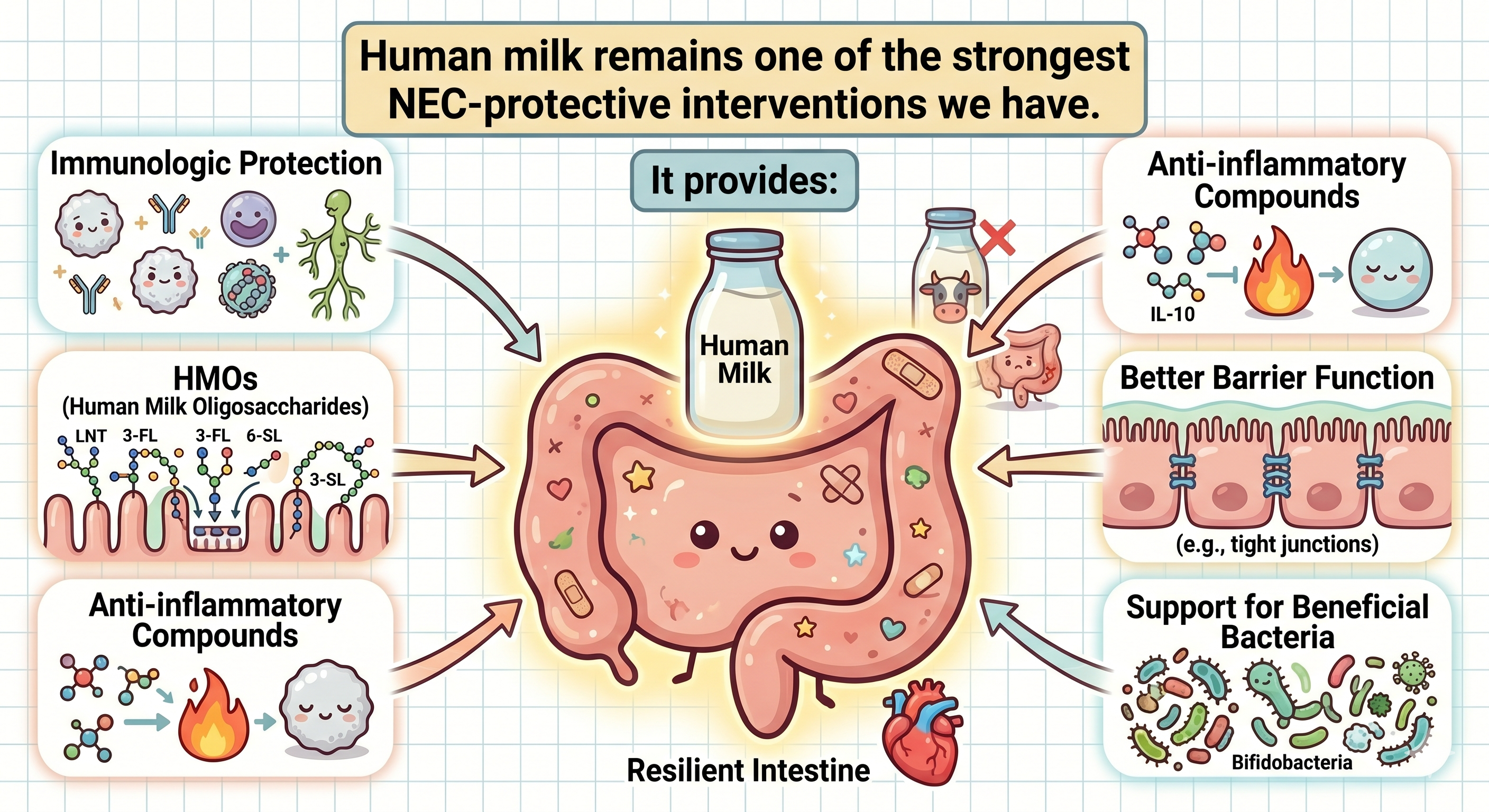
Recent research has highlighted the possible protective role of specific HMOs, including disialyllacto-N-tetraose (DSLNT), which has shown promising associations with reduced NEC risk. These oligosaccharides essentially act as prebiotics, feeding the beneficial organisms that help protect the preterm gut.
Human milk-based fortifiers are also associated with lower NEC risk compared to cow's milk-based fortifiers in very small infants.
For NICU nurses, supporting lactation is essential evidence-based NEC prevention.
That includes:
Early pumping education and establishing supply
Skin-to-skin care
Protecting and sustaining milk supply throughout the hospitalization
Encouraging pumping frequency
Supporting donor milk programs
Helping families understand why human milk matters
Oral Immune Therapy: A Nurse-Led Practice With Real Impact
If you're not already familiar with oral immune therapy (OIT), also called oropharyngeal administration of colostrum, this is one of the most exciting nurse-led interventions in the NICU and it has direct relevance to NEC prevention. I get really excited to talk about it!
OIT involves applying small amounts of the mother's own colostrum or breast milk to the oral mucosa of preterm infants who are not yet able to receive enteral feeds. The mucosa-associated lymphoid tissue (MALT) in the oropharynx can absorb the immunologic components of human milk directly even when the gut isn't ready.
Why It Matters for NEC
The preterm gut is highly vulnerable in the days before enteral feeding is established. During this window, OIT may help by:
Delivering secretory IgA, lactoferrin, lysozyme, and other immune factors directly to mucosal surfaces
Priming the mucosal immune system before feeds begin
Providing growth factors and anti-inflammatory cytokines that support intestinal maturation
Supporting early microbiome colonization through exposure to the mother's milk microbiota
Several studies have shown associations between OIT and reduced rates of sepsis, lower NEC incidence, and improved feeding tolerance in very preterm and very low birth weight infants.
Technique Matters
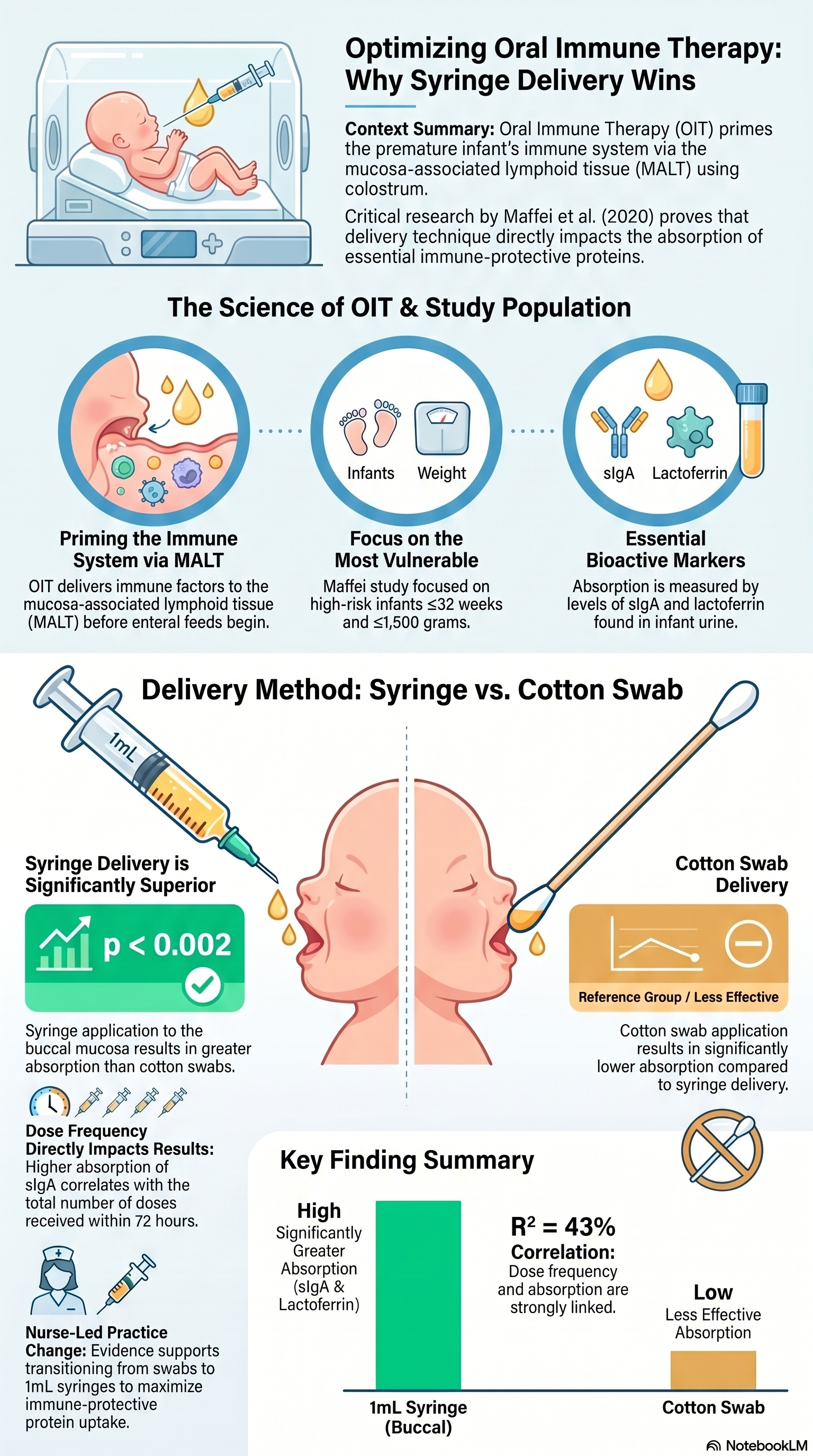
A study by Maffei et al. published in the Journal of Perinatology is essential knowledge for every NICU nurse doing OIT.
The study enrolled infants ≤32 weeks and ≤1,500 grams and measured sIgA and lactoferrin in infant urine as a marker of mucosal absorption. Importantly, all sampling was done before enteral feeds were initiated, so any absorbed immune proteins were attributable solely to OIT. They compared two delivery methods: cotton swab versus 1mL syringe applied directly to the buccal mucosa.
The findings were striking: urinary sIgA and lactoferrin were significantly greater in infants who received colostrum by syringe compared to swab (p < 0.002). And dose frequency mattered too! Urinary sIgA correlated with the total number of doses received in 72 hours (R² = 43%, p < 0.01).
What this means practically:
Syringe delivery to the buccal mucosa is superior to cotton swab for maximizing absorption of immune-protective proteins
More doses = more absorption: consistency and frequency of administration are clinically meaningful
Early initiation may contribute to a more diverse tracheal microbiome, suggesting effects that extend beyond the gut
If your unit is still using cotton swabs for OIT, this paper is worth bringing to your educator, CNS, or nursing leadership. The technique your nurses use influences how much of colostrum's immune protection actually reaches the infant's system.
The Nursing Role in OIT
This is a practice that nurses champion, teach, and implement. It requires:
Early colostrum collection education: helping mothers understand the importance of collecting even tiny drops of colostrum in the first hours and days after delivery
Technique: delivering colostrum every 2–3 hours as available per unit protocol
Using the 1mL syringe is more effective compared to a swab
Documentation and advocacy: ensuring OIT is initiated promptly and consistently, even for the most critically ill infants who are NPO
One of the most powerful things about OIT is that it gives families something meaningful to do for their baby during some of the hardest days of their lives. It is a tangible, evidence-informed act of care at a time when parents often feel helpless.
For nurses studying for the RNC-NIC or CCRN: know that OIT is increasingly featured in neonatal nursing literature as part of human milk promotion and neuroprotective developmental care bundles. Understanding the mechanism (mucosal immune priming through MALT) helps you connect the practice to the broader science of why human milk matters so much in the preterm population.
Standardized Feeding Protocols Matter
A common quality improvement strategy in NEC prevention is the use of standardized feeding protocols. Research shows that protocol-driven feeding approaches for infants under 1,500 grams can reduce NEC incidence by approximately 67%.
These protocols help reduce:
Wide provider-to-provider variation
Unnecessary feeding interruptions
Inconsistent advancement strategies
Delayed feeding progression
Recent literature also challenges some older practices many NICU nurses were taught years ago.
Routine gastric residual checks do not prevent NEC
Several studies suggest routine residual aspiration in asymptomatic infants may not improve outcomes and could potentially disrupt feeding progression unnecessarily. This is an area where NICU practice continues to evolve and it's worth knowing what your unit's current protocol says and why. Do you still routinely aspirate gastric residuals? I was so happy to see at the ONE conference almost no hands went up when I asked that question.
"Slow feeding" is not a prevention strategy
The evidence does not support delayed feeding initiation or prolonged slow advancement as a strategy to prevent NEC. Following your unit's standardized protocol is the strongest evidence-based approach.
Antibiotic Stewardship Is NEC Prevention
One important finding in recent NEC research is that prolonged antibiotic exposure may actually increase NEC risk because antibiotics alter the developing microbiome.
Research suggests prolonged empirical antibiotics:
Reduce microbial diversity
Promote dysbiosis and Proteobacteria overgrowth
Increase intestinal permeability
Alter immune signaling
This does not mean antibiotics are unnecessary. Septic preterm infants absolutely need prompt treatment. But it does reinforce the importance of reassessing antibiotics at 36-48 hours, avoiding prolonged "just in case" exposure when cultures remain negative and the clinical picture is improving, and recognizing that antibiotic stewardship is not just about resistance. It is also about protecting the microbiome that protects these babies.
Similarly, routine antacid and acid-suppressing medications should not be used without clear clinical indication, as these also alter the gut environment in ways associated with increased NEC risk.
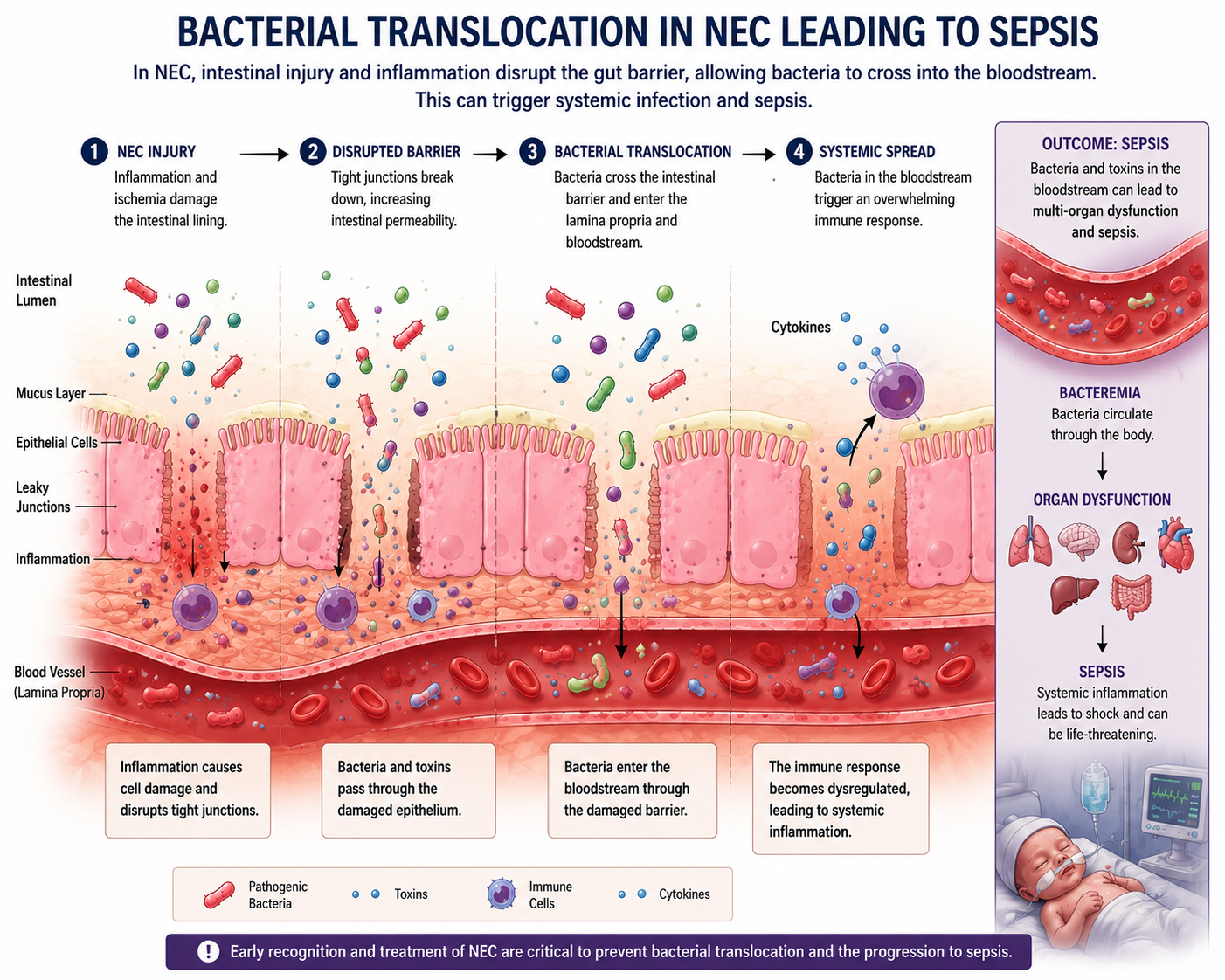
NEC and Sepsis Are Deeply Connected
As NEC progresses, bacterial translocation through the damaged intestinal barrier can lead to overwhelming sepsis. This is why babies with NEC can deteriorate so quickly.
NICU nurses are often the first to notice:
Temperature instability
Increased apnea and bradycardia events
Worsening perfusion
Increasing abdominal discoloration or distention
Lethargy and tone changes
Metabolic acidosis
Escalating respiratory support needs
Early recognition and escalation truly matter and validated tools like GutCheckNEC and structured SBAR communication can help when NEC is suspected. Frequent serial abdominal assessments, monitoring for systemic signs consistent with Bell staging, and knowing when to escalate are core nursing competencies that directly influence outcomes.
Biomarkers and Early Detection Are a Major Research Focus
One of the biggest challenges in NEC is that early symptoms are nonspecific. Researchers are actively investigating biomarkers that may improve early detection.
A 2026 systematic review identified several promising markers, based on the timing of their appearance:
Prior to Diagnosis: Neutrophil CD64+ and certain VOCs (like Pent-1-ene-3-one) are identified as the strongest early predictors
Day of Diagnosis: Faecal calprotectin (97.14% sensitivity, 100% specificity) and serum calprotectin (100% sensitivity, 96.4% specificity) were among the most accurate established markers
Biomarker Panels: A panel consisting of urine proteins Cystatin C (CST3), PEDF, and Retinol Binding Protein 4 (RET4) demonstrated high sensitivity (96%) and specificity (90%)
Medical vs. Surgical Differentiation: To identify infants requiring surgery, the review found that PCT (Procalcitonin), Factor XIII, and the RELM-β protein provided high discriminatory power
Machine learning models are also being applied to improve risk stratification and enhance imaging-based early detection though these require large, diverse datasets and significant clinical validation before they become standard tools.
The challenge with biomarkers remains that many look promising in research studies but have not yet been validated broadly enough for routine bedside use. For now, serial bedside assessment by experienced NICU nurses remains critically important. That clinical intuition (when you sense something is "off" before the labs confirm it) is not imagined. It reflects pattern recognition built from thousands of assessments, and it saves lives.
The Future of NEC Research
Current NEC research is exploring:
Microbiome-directed therapies, including specific probiotic strains and SCFA supplementation
Bacteroides fragilis as a potential protective microorganism
TLR4 inhibitors and other immune-targeted approaches
β-glucan for barrier protection and microbiome modulation
Stem cell therapies
AI-assisted risk prediction
Precision medicine approaches based on the idea that NEC likely represents a spectrum of related disorders rather than a single disease
Despite these advances, prevention remains centered around fundamentals:
Human milk
Careful, standardized feeding practices
Antibiotic and medication stewardship
Microbiome-protective care
Early recognition and skilled nursing assessment
What NICU Nurses Should Take Away
NEC is one of the clearest examples of how bedside nursing care directly influences outcomes.
The research continues to evolve… from TLR4 pathways to dysbiotic microbiomes to metabolite disruptions, but the nursing impact remains enormous and consistent across the literature.
NICU nurses help prevent NEC when they:
Advocate for human milk and support lactation at every opportunity
Follow evidence-based, standardized feeding protocols
Perform vigilant, serial abdominal assessments
Recognize subtle early changes and escalate concerns promptly
Practice antibiotic and medication stewardship
Educate and partner with families. Including teaching parents NEC warning signs and inviting them to speak up with concerns
Protect the microbiome through every care decision, from milk choice to medication use to oral immune therapy and skin-to-skin care
Perhaps most importantly: the experienced NICU nurse who recognizes NEC before the disease fully declares itself is not lucky. They are skilled. That pattern recognition, built over years at the bedside, is one of the most powerful diagnostic tools we have and no biomarker panel has replaced it yet.
References
Duess, J. W., Sampah, M. E., Lopez, C. M., Tsuboi, K., Scheese, D. J., Sodhi, C. P., & Hackam, D. J. (2023). Necrotizing enterocolitis, gut microbes, and sepsis. Gut Microbes, 15(1). https://doi.org/10.1080/19490976.2023.2221470
Faizan, M. A., Khalid, I., Yeo, A., et al. (2026). Necrotising enterocolitis biomarkers: A systematic review. Frontiers in Pediatrics, 13, 1652566. https://doi.org/10.3389/fped.2025.1652566
Hu, X., Liang, H., Li, F., et al. (2024). Necrotizing enterocolitis: Current understanding of the prevention and management. Pediatric Surgery International, 40(32). https://doi.org/10.1007/s00383-023-05619-3
Ishiyama, A., Jang, H., Dintaman, J., et al. (2025). Necrotizing enterocolitis: A comprehensive review on Toll-like receptor 4-mediated pathophysiology, clinical, and therapeutic insights. Biomedicines, 13. https://doi.org/10.3390/biomedicines13092288
Kaplina, A., Kononova, S., Zaikova, E., et al. (2023). Necrotizing enterocolitis: The role of hypoxia, gut microbiome, and microbial metabolites. International Journal of Molecular Sciences, 24(3), 2471. https://doi.org/10.3390/ijms24032471
Maffei, D., & Schanler, R. (2017). Human milk is the feeding strategy to prevent necrotizing enterocolitis! Seminars in Perinatology, 41(1), 36–40. https://doi.org/10.1053/j.semperi.2016.09.016
Monzon, N., Kasahara, E., Gunasekaran, A., Burge, K., & Chaaban, H. (2023). Impact of neonatal nutrition on necrotizing enterocolitis. Seminars in Pediatric Surgery, 32(3), 151305. https://doi.org/10.1016/j.sempedsurg.2023.151305
Roberts, A., Younge, N., & Greenberg, R. (2024). Neonatal necrotizing enterocolitis: An update on pathophysiology, treatment, and prevention. Pediatric Drugs, 26, 259–275. https://doi.org/10.1007/s40272-024-00626-w
Sha, C., Sander, W. R., Bass, K., et al. (2025). Necrotizing enterocolitis: What's new and what's next? International Journal of Molecular Sciences, 26(19), 9660. https://doi.org/10.3390/ijms26199660
Singh, D. K., Miller, C. M., Orgel, K. A., et al. (2023). Necrotizing enterocolitis: Bench to bedside approaches and advancing our understanding of disease pathogenesis. Frontiers in Pediatrics, 10, 1107404. https://doi.org/10.3389/fped.2022.1107404
Cifuentes, M., Chapman, J., & Stewart, C. (2024). Gut microbiome derived short chain fatty acids: Promising strategies in necrotising enterocolitis. Current Research in Microbial Sciences, 6. https://doi.org/10.1016/j.crmicr.2024.100219
Chen, Z., Chen, H., Huang, W., et al. (2024). Bacteroides fragilis alleviates necrotizing enterocolitis through restoring bile acid metabolism balance using bile salt hydrolase and inhibiting FXR-NLRP3 signaling pathway. Gut Microbes, 16. https://doi.org/10.1080/19490976.2024.2379566
Allana, A., Bashir, S., & Hand, I. (2025). Quality improvement project to improve adherence to best practices to decrease incidence of necrotizing enterocolitis in preterm infants. Children, 12. https://doi.org/10.3390/children12020176
Ramadan, O., Alruwaili, M., Alruwaili, A., et al. (2024). Nursing practice of routine gastric aspiration in preterm infants and its link to necrotizing enterocolitis: Is the practice still clinically relevant? BMC Nursing, 23. https://doi.org/10.1186/s12912-024-01994-x
Rodriguez, N. A., Meier, P. P., Groer, M. W., Zeller, J. M., Engstrom, J. L., & Fogg, L. (2010). A pilot study to determine the safety and feasibility of oropharyngeal administration of own mother's colostrum to extremely low-birth-weight infants. Advances in Neonatal Care, 10(4), 206–212. https://doi.org/10.1097/ANC.0b013e3181e94133
Seigel, J. K., Smith, P. B., Ashley, P. L., Cotten, C. M., Herbert, C. C., King, B. A., & Bidegain, M. (2013). Early administration of oropharyngeal colostrum to extremely low birth weight infants. Breastfeeding Medicine, 8(6), 491–495. https://doi.org/10.1089/bfm.2013.0025
Maffei, D., Brewer, M., Codipilly, C., Torres, C., Weinberger, B., Schanler, R. J., & Angert, R. M. (2020). Early oral colostrum administration in preterm infants. Journal of Perinatology, 40(2), 284–289. https://doi.org/10.1038/s41372-019-0556-x

December 2023 Certification Review Webinar
NICU Certification Review



Ready to kickstart your journey to becoming a certified NICU nurse?
Look no further!
Grab my FREE E-Book packed with essential study and test-taking strategies for the RNC-NIC.
In the E-Book I give you the resources you need including the link to access the candidate guide, several types of books to study from, some of my favorite strategies, an outline of the content you should review, and a blank calendar for you to make your study plan!
Frequently Asked Questions About the RNC-NIC exam

What is the RNC-NIC?
The RNC-NIC is a competency-based exam that tests the specialty knowledge of nurses in the United States & Canada who care for critically ill newborns and their families.
The RNC-NICU is a nationally recognized certification that recognizes the registered nurse for their specialty knowledge and skill.

Who can take the RNC-NIC exam?
Nurses can take this exam after a minimum of two years experience in the NICU caring for critically ill newborns and their families.

Which books should I use?
I'm glad you asked! There are many excellent books to help you prepare for the RNC-NIC, I gathered ande describe each of them for you in my FREE e-book.
Is there a course to help me study?
Yes! Many hospitals host their own certification course and there are a few online courses. See my RNC-NIC test taking tips E Book for more information
What happens if I don't pass the exam?
If you don't pass the exam on your first try you can try again after 90 days. You will have to reapply after 90 days and pay a retest fee. There is no limit to the number of times you can take the exam (however a candidate can only sit for the exam twice per year).
Can I make more money if I take the RNC-NIC exam and get certified?
Yes! Many hospitals provide a raise or a bonus for nurses with specialty certifications. Hospitals also typically hire at a higher base salary when nurses have a certification.

Find me @amandasnicued on these channels or Email me
hey nurses don't miss out
© Copyright 2024. AmandasNICUEd. All rights reserved. | Terms & Conditions | Privacy Policy Contact: [email protected]

